Antiviral test : COVID-19
◯ Sample submission: November 4, 2020 / Response date: December 28, 2020
◯ Client: Presence, Inc.
◯ Test item: Antiviral activity test
◯ Test method: ISO21702 / Measurement of antiviral activity on plastics and other non-porous surfaces
◯ Test organization: QTEC, Japan Textile Products Quality Technology Center, Kobe Testing Center, Microorganism Testing Laboratory
Test Summary
◯ Test virus: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) / NIID isolate:
JPN/TY!WK-521 (distributed by the National Institute of Infectious Diseases)
・Host cell:VeroE6/TMPRSS2 JCRB1819
・Cytoplasmic drop : Dulbecco’s modified Eagle’s medium (low-glucose) ; DMEM
(SIGMA, Cat#D6046) Minimum Essential Medium Eagle ; EMEM (SIGMA, Cat#-M4655)
・Fetal Bovine Serum : Fetal Bovine Serum (FBS) (SIGMA, Cat#l 73012)
・Sealing film: Polyethylene film
・Control sample : GlossWell #360 Type Anti-Viral (Unprocessed products)
・Test sample : GlossWell #360 Type Anti-Viral (Processed products)
・Purification of test specimen: Not performed
・Inoculation of test virus suspension: 0.4ml
・Test condition: Action temperature: 25°C
・Test condition: Action time: 24 hours (The virus infection titer was also measured immediately after inoculation for the control sample.
・Washing solution: SCDLP diluted 10 times in DMEM containing 2% FBS
・Infection titer measurement method: Plaque measurement met
Test Operation
1 ) Main Test :
1. Infect host cells with the virus, add EMEM and incubate at 37°C for a predetermined period of time, then centrifuge at 4°C, l,000xg for 15 minutes, and the supernatant is used as the test virus suspension.
2. Dilute the virus suspension obtained in 1. 10-fold with sterile distilled water to 1 ~sx101 PFU/mL, and use this as the test virus suspension.
3. Place each specimen (50 mm x 50 mm) on the bottom of a sterile Petri dish with the processed side up, and inoculate with 0.4 ml of the test virus suspension.
4. Cover the petri dish with an adhesive film (40 mm x 40 mm) and press down lightly so that the test virus suspension is spread over the entire film.
5. Cover the petri dish with the lid.
6. After dying at 25°C for 24 hours at 90%RH or higher, add 10 mL of washing-up liquid to each test sample.
7. Scrub the surface of each test specimen and the dense cling film to wash out the virus.
8. Dilute the washout solution 10-fold using DMEM containing 2% FBS.
9. Determine the viral infection titer by the plaque measurement method.
2 ) Host cell verification test :
2 )-1 Cytotoxicity Confirmation
1. Add l0mL of washing-up liquid to each test sample, and perform the washing-up procedure as in the main test.
2. Make a 10-fold dilution of the washout solution using DMEM containing 2% FBS.
3. Stain the cells as in the plaque measurement method to check for cytotoxicity.
2 )-2 Susceptibility testing of cells to viruses
1. Add 10mL of washing-up liquid to each test sample and perform the washing-up procedure as in the main test.
2. Dilute the washout solution 10-fold using DMEM containing 2% FBS.
3. Take 5 mL of the solution in 2. above into a sterile test tube.
4. Prepare a test virus suspension at 4~6×104 PFU/ml using EMEM, and add 0.05 ml of the suspension to the washout solution in 2.
5. Allow to stand at 25°C for 30 minutes.
6. Measure the virus infection titer by the plaque measurement method and confirm the susceptibility of the cells to the virus by measuring the virus infection titer per ml of washout solution.
Test Results
1 ) Main Test :
・Test virus: SARS-CoV-2 NIID isolate; JPN/TY!WK-521 (distributed by the National Institute of Infectious Diseases)
・Test virus suspension concentration: 1.2 x 107 PFU/ml
| Specimen | Virus infection titer (PFU/cm2) (Note 2) Logarithmic value for normal use | Virus infection titer (PFU/cm2) (Note 2) log-average normalized value | Antiviral activity value 【R】 (Note 2) |
|---|---|---|---|
| GlossWell #360 Type Anti-Viral (Unprocessed) (Note 1) Immediately after inoculation【U0】 | n1 / 5.52 | 5.53 | |
| same as above | n2 / 5.52 | 5.53 | |
| same as above | n3 / 5.55 | 5.53 | |
| GlossWell #360 Type Anti-Viral (Unprocessed product) (Note 1) After leaving for 24 hours【Ut】 | n1 / 5.04 | 5.04 | |
| same as above | n2 / 5.03 | 5.04 | |
| same as above | n3 / 5.06 | 5.04 | |
| GlossWell #360 Type Anti-Viral (Processed product) After leaving for 24 hours【At】 | n1 <1.80 | <1.80 | |
| same as above | n2 <1.80 | <1.80 | |
| same as above | n3 <1.80 | <1.80 | ≧3.2 [ Numeric Description ] |
The antiviral activity value ≧3.2 means that the antiviral activity value after 24 hours is 99.9% or more than 1/1000.
※ The antiviral activity value that is considered acceptable by ISO 21072 is ≧2.0, so the results of this test far exceed the acceptable value.
(Note 1) : GlossWell #360 Type Anti-Viral (unprocessed) (provided by the client) was used as a control sample.
(Note 2) PFU : plaque forming units.
(Note 3) Antiviral activity value R= Ut -At
2 ) Host cell verification test
・Test virus: SARS-CoV-2 NIID isolate; JPN/TY/WK-521 (distributed by the National Institute of Infectious Diseases)
・Test virus suspension concentration: 4.9 x 104PFU/ml
| Specimen | 2 ) -1 Presence of cytotoxicity | 2 ) -2 Confirmation of cell susceptibility to the virus. Virus infection titer (PFU/ml) (Note 2) Logarithmic mean value for normal use | Determination of test validity |
|---|---|---|---|
| GlossWell #360 Type Anti-Viral (Unprocessed) (Note 1) | non- | 【 Su 】 2.68 | establishment |
| GlossWell #360 Type Anti-Viral (Processed product) | non- | 【 Su 】 2.69 | establishment |
| Negative control (Note 4) | non- | 【 Sn 】 2.67 |
(Note 4) A solution of SCDLPs diluted 10-fold in DMEM containing 2% FBS was used as a negative control.
Conditions for completion of the test
2-1 ) Cytotoxicity: None
2-2 ) Confirmation of cell susceptibility to the virus: | Sn – Su | ≤ 0.5 and | Sn – S1 | ≤ 0.5
Reference Information
◯ Real-time RT-PCR measurement of virus suspensions subjected to this test.
・Test virus: SARS-CoV-2NIID isolate; JPN/TY/WK.-521 (distributed by the National Institute of Infectious Diseases)
・Virus suspension wastiness: >lOSPFU/ml
・Real-time PCR equipment: Thermal Cycler Dice Real Time SysteM 3 (TaKaRa)
・Detection Kit: SARS-CoV-2Detection Kit -Nl set- (Code NCV-301; Lot# 038200)
(TOYOBO CO.,LTD. Biotech support Department)
◯ Measurement results
The amplification of viral RNA was confirmed by the real-time RT-PCR measurement results (Fig. l).
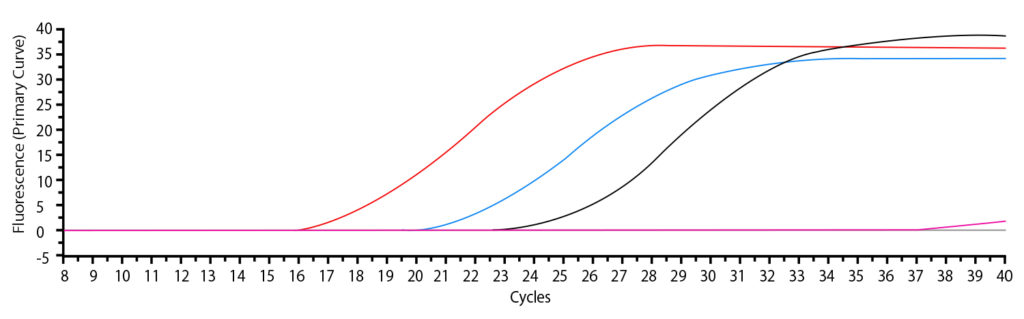
◯ Graph: red line (102-fold dilution of viral suspension flooding in PBS)
◯ Graph: Blue line (103-fold dilution of virus suspension wastage in PBS)
◯ Graph: black line (104-fold dilution of virus suspension wastage in PBS)
◯ Graph: pink line (Negative control; EMEM)
Testing organization: Microbiological Testing Laboratory, Kobe Testing Center, Japan Textile Quality Technology Center
BL HY-COATER
PRESENCE Co., Ltd. / BADLAND
Post : 230-0073
2-39-45 Shishigaya Tsurumi-ku Yokohama Kanagawa JAPAN
TEL +81-45-717-7026 / FAX +81-45-717-7027 / e-mail: info@badland.net


Comment On Facebook